Fda Protocol Template
Fda Protocol Template - Web clinical trial protocols should include a clear description of trial design and patient selection criteria as well as description of. Web this page includes seven different protocol templates for developing a variety of different new research protocols. Web 138 rows clinical trials guidance documents. Web 15 rows comparison of fda, epa, oecd glp protocol & conduct; Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. Office of generic drugs and office of. Web the fda staff responsible for this guidance as listed on the title page. Web fda updates the clinical protocol template. Center for drug evaluation and research, office of regulatory policy this template. Guidance documents listed below represent the agency's. Guidance documents listed below represent the agency's. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. The national institutes of health (nih) and food and drug administration (fda) developed a clinical. (thursday, january 19, 2023) the fda recently released an. Protocol concurrence will be issued solely. Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Web click the thumbnail to access a free template. Office of generic drugs and office of. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national. Web 15 rows comparison of fda,. Web clinical trial protocols should include a clear description of trial design and patient selection criteria as well as description of. The draft guidance has been. The national institutes of health (nih) and food and drug administration (fda) developed a clinical. Web to set this template's initial visibility, the |state= parameter may be used: Web the fda staff responsible for. Web this template provides the food and drug administration’s (fda) current recommendations concerning what data. The national institutes of health (nih) and food and drug administration (fda) developed a clinical. The electronic protocol writing tool aims to facilitate the development of. Guidance documents listed below represent the agency's. (thursday, january 19, 2023) the fda recently released an. Web to set this template's initial visibility, the |state= parameter may be used: Office of generic drugs and office of. Web 20 drug administration (fda) and sponsors or applicants relating to the development and review 21 of drug or biological drug. Web 138 rows clinical trials guidance documents. Center for drug evaluation and research, office of regulatory policy this template. Web 15 rows comparison of fda, epa, oecd glp protocol & conduct; Web to set this template's initial visibility, the |state= parameter may be used: Web center for drug evaluation and research mapp 5220.8 rev. Office of generic drugs and office of. The draft guidance has been. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. The electronic protocol writing tool aims to facilitate the development of. Web center for drug evaluation and research mapp 5220.8 rev. Web click the thumbnail to access a free template. Web the fda staff responsible for this. Center for drug evaluation and research, office of regulatory policy this template. Web developed jointly by nih and the food and drug administration (fda), the protocol template provides a standard. Web this template provides the food and drug administration’s (fda) current recommendations concerning what data. Web this page includes seven different protocol templates for developing a variety of different new. Web 15 rows comparison of fda, epa, oecd glp protocol & conduct; Center for drug evaluation and research, office of regulatory policy this template. Web 138 rows clinical trials guidance documents. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. Web center for drug evaluation and. Web developed jointly by nih and the food and drug administration (fda), the protocol template provides a standard. Center for drug evaluation and research, office of regulatory policy this template. Web this page includes seven different protocol templates for developing a variety of different new research protocols. The national institutes of health (nih) and food and drug administration (fda) developed. The electronic protocol writing tool aims to facilitate the development of. Web the fda staff responsible for this guidance as listed on the title page. Web to set this template's initial visibility, the |state= parameter may be used: Office of generic drugs and office of. Protocol concurrence will be issued solely based upon the information you provide in the qbr template. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. The national institutes of health (nih) and food and drug administration (fda) developed a clinical. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national. Web clinical trial protocols should include a clear description of trial design and patient selection criteria as well as description of. Web this page includes seven different protocol templates for developing a variety of different new research protocols. The draft guidance has been. Web 15 rows comparison of fda, epa, oecd glp protocol & conduct; (thursday, january 19, 2023) the fda recently released an. Web 20 drug administration (fda) and sponsors or applicants relating to the development and review 21 of drug or biological drug. Web developed jointly by nih and the food and drug administration (fda), the protocol template provides a standard. Guidance documents listed below represent the agency's. Format and content of a rems document: Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Web center for drug evaluation and research mapp 5220.8 rev. Web developed jointly by nih and the food and drug administration (fda), the protocol template provides a standard. Web this page includes seven different protocol templates for developing a variety of different new research protocols. Web the fda staff responsible for this guidance as listed on the title page. Web 15 rows comparison of fda, epa, oecd glp protocol & conduct; Web 138 rows clinical trials guidance documents. Web 20 drug administration (fda) and sponsors or applicants relating to the development and review 21 of drug or biological drug. Web clinical trial protocols should include a clear description of trial design and patient selection criteria as well as description of. Center for drug evaluation and research, office of regulatory policy this template. Web this protocol template aims to facilitate the development of two types of clinical trials involving human. Web this clinical trial protocol template is a suggested format for phase 2 and 3 clinical trials funded by the national institutes of. Web this template provides the food and drug administration’s (fda) current recommendations concerning what data. Web click the thumbnail to access a free template. Protocol concurrence will be issued solely based upon the information you provide in the qbr template. The draft guidance has been. Format and content of a rems document: Web to set this template's initial visibility, the |state= parameter may be used:Clinical Trial Protocol
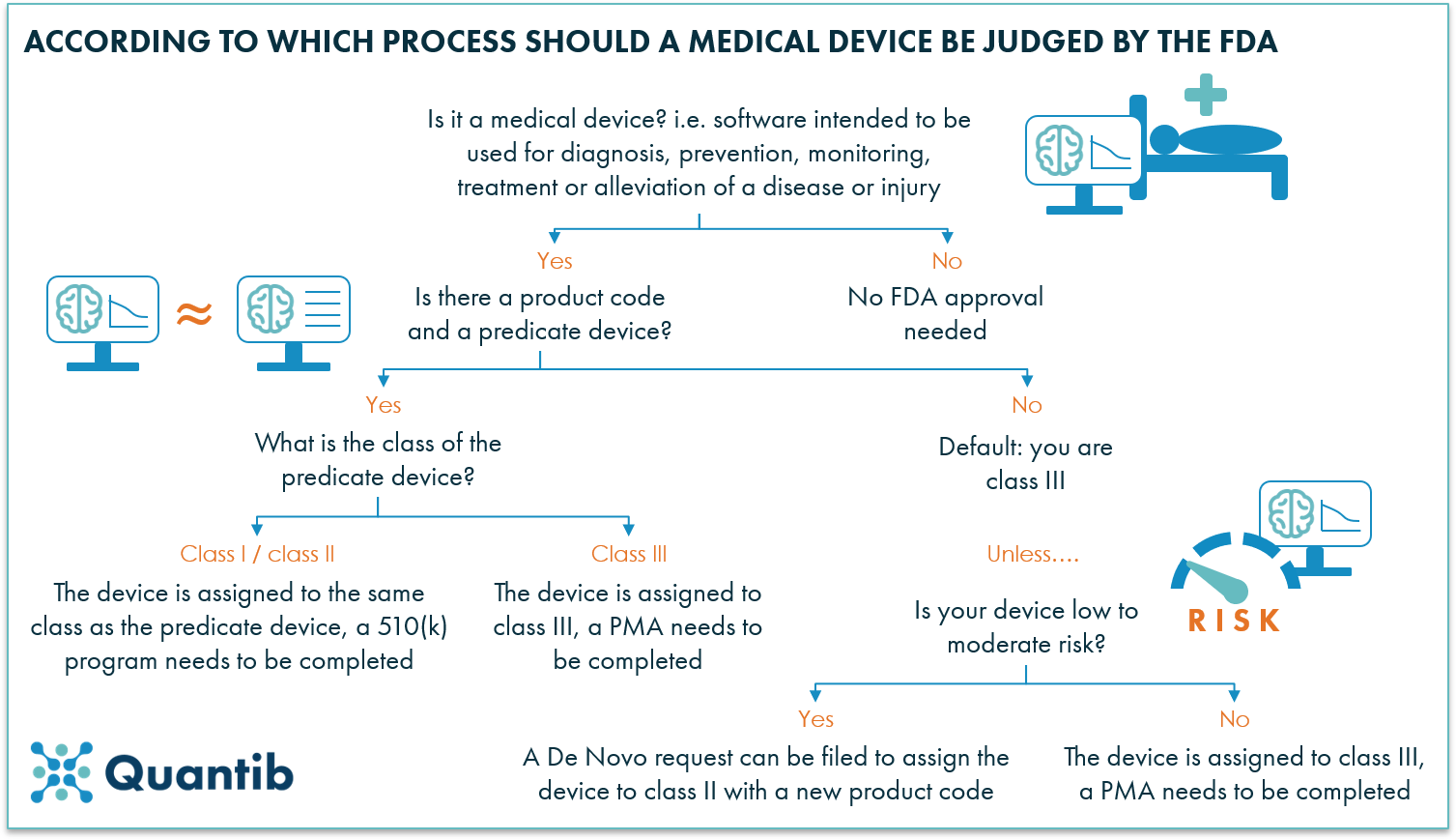
A 101 guide to the FDA regulatory process for AI radiology software
Stability Study Protocol Template williamsonga.us
PROCESS VALIDATION SOP Template MD46 GMP, QSR & ISO Compliance
Protocol Template 05Feb2016 508 Clinical Trial Food And Drug
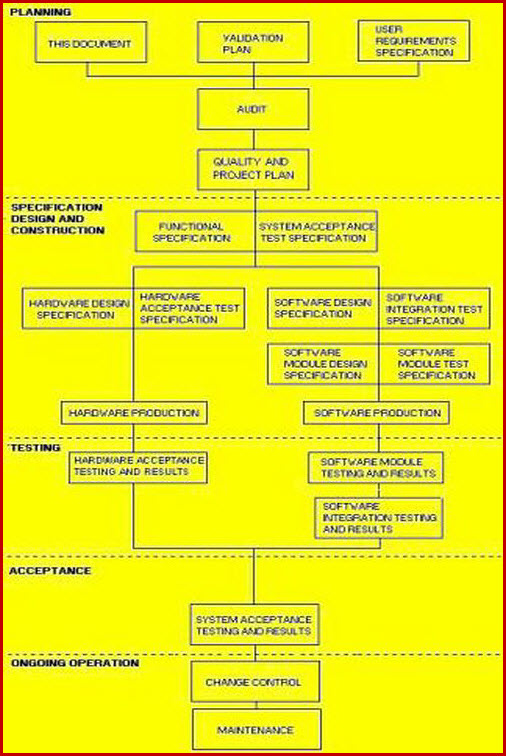
Validation Master Plan FDA EU WHO Pharma Meddevice Bio
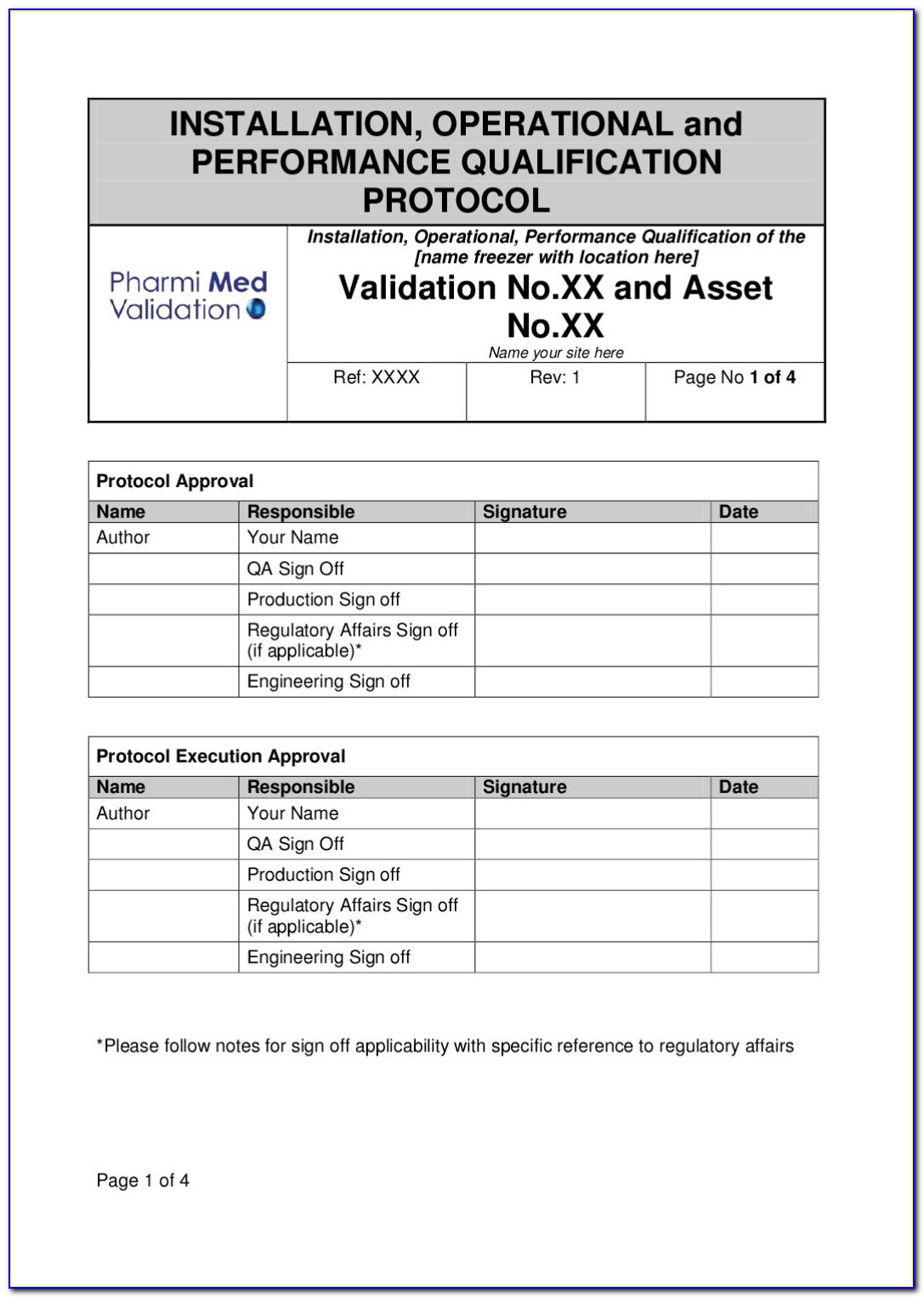
Iq Oq Pq Software Validation Templates
Retail policy and procedure manual template
Three steps to plan for the FDA’s new food label rules 20161018
FDA Software Validation 2022 Guide, Checklist & Template
Guidance Documents Listed Below Represent The Agency's.
The Electronic Protocol Writing Tool Aims To Facilitate The Development Of.
Web This Clinical Trial Protocol Template Is A Suggested Format For Phase 2 And 3 Clinical Trials Funded By The National Institutes Of.
Web Fda Updates The Clinical Protocol Template.
Related Post: