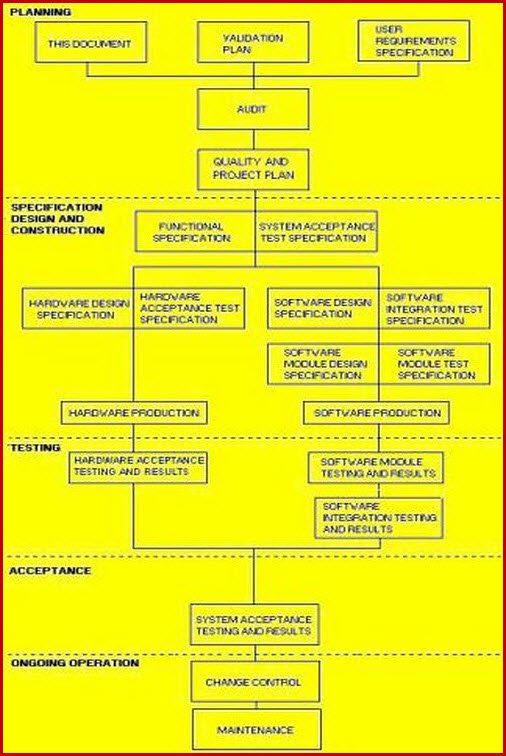
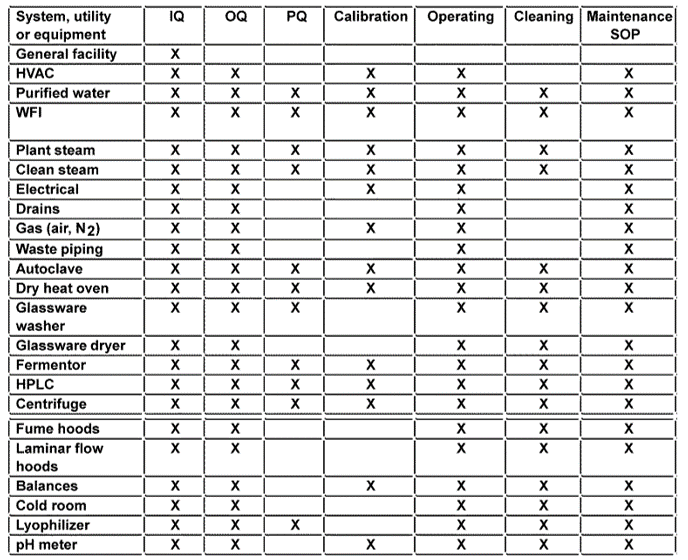
Master Validation Plan Template
Master Validation Plan Template - Web download now validation master plan download now calibration and validation plan download now software validation plan. Web pharmacy manufacturing unit validation master plan (vpm). Web what is a validation master plan template? Web validation master plan template document control details this will include details such as vmp reference number, version. Web download now of 26 document no. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. To see the complete list of the most popular validation templates, click here. This template is a tool for creating a customized plan for validating a product, system, or process. Web three (3) options to create a validation master plan. The purpose of the validation master plan is to document the. Web the guidance even includes templates for a master validation plan, iq, oq, and pq. Web pharmacy manufacturing unit validation master plan (vpm). Web this template describes the information that needs to be presented in a validation master plan and provides examples. You can create a great protocol, using a template. Web a validation master plan (vmp) drives a structured. 5.2.7 for large projects involving many materials,. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. Web three (3) options to create a validation master plan. Web a validation master plan (vmp) drives a structured approach to validation projects that will allow you to address many. Web download. Web use this template to: To see the complete list of the most popular validation templates, click here. Web download now validation master plan download now calibration and validation plan download now software validation plan. Web the validation master plan is a summary of validation strategy. It can include information such as the scope of work, timeline, resources needed, risk. Your pvp should contain the following elements:. Web what is a validation master plan template? Web the purpose of the validation master plan template (vmp) is to describe the organization’s overall strategy, approach, and responsibilities for. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. The purpose of. Web 1 objectives of the document the cleaning processes must be validated to confirm the efficiency of the method. Web what is a validation master plan template? You can download a free sample of a validation master plan template in.pdf format. Web this template describes the information that needs to be presented in a validation master plan and provides examples.. You can create a great protocol, using a template. Web the guidance even includes templates for a master validation plan, iq, oq, and pq. Web a validation master plan (vmp) drives a structured approach to validation projects that will allow you to address many. Web pharmacy manufacturing unit validation master plan (vpm). Web three (3) options to create a validation. Web the validation master plan is a summary of validation strategy. Web 1 objectives of the document the cleaning processes must be validated to confirm the efficiency of the method. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. The cleaning validations are done for. The purpose of. Web the guidance even includes templates for a master validation plan, iq, oq, and pq. Web this template describes the information that needs to be presented in a validation master plan and provides examples. Web the technical storage or access is required to compose employee profiles to send advertising, or to track the. Web download now validation master plan download. Validation master plan (vmp) for the pharma co., effective date: Web three (3) options to create a validation master plan. Web the validation master plan serves as a roadmap that helps to set the course, justifying the strategy, outlined the. Your pvp should contain the following elements:. Web download now of 26 document no. You can create a great protocol, using a template. This template is a tool for creating a customized plan for validating a product, system, or process. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. Web pharmacy manufacturing unit validation master plan (vpm). Web download now of 26. This template is a tool for creating a customized plan for validating a product, system, or process. Web validation master plan is a document, which describes our company’s intentions and the methods which are related with validation. You can download a free sample of a validation master plan template in.pdf format. Web a validation master plan (vmp) outlines the principles involved in the qualification of a facility, defining the areas and systems to be validated,. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. Web pharmacy manufacturing unit validation master plan (vpm). It can include information such as the scope of work, timeline, resources needed, risk assessment criteria, quality control procedures, and acceptance criteria. The purpose of the validation master plan is to document the. Web three (3) options to create a validation master plan. Your pvp should contain the following elements:. Web validation master plan template document control details this will include details such as vmp reference number, version. Web the technical storage or access is required to compose employee profiles to send advertising, or to track the. Web the purpose of the validation master plan template (vmp) is to describe the organization’s overall strategy, approach, and responsibilities for. 5.2.7 for large projects involving many materials,. Web download now validation master plan download now calibration and validation plan download now software validation plan. Web a validation master plan (vmp) drives a structured approach to validation projects that will allow you to address many. You can create a great protocol, using a template. Web 1 objectives of the document the cleaning processes must be validated to confirm the efficiency of the method. Validation master plan (vmp) for the pharma co., effective date: Web this template describes the information that needs to be presented in a validation master plan and provides examples. Web pharmacy manufacturing unit validation master plan (vpm). Validation master plan (vmp) for the pharma co., effective date: 5.2.7 for large projects involving many materials,. Web the validation master plan is a summary of validation strategy. Web a validation master plan (vmp) outlines the principles involved in the qualification of a facility, defining the areas and systems to be validated,. Web the validation master plan is designed to provide a planned and systematic framework within which all validation activities will occur. Web use this template to: Web download now validation master plan download now calibration and validation plan download now software validation plan. Web 1 objectives of the document the cleaning processes must be validated to confirm the efficiency of the method. Web download now of 26 document no. Web validation master plan template document control details this will include details such as vmp reference number, version. Web three (3) options to create a validation master plan. Web validation master plan is a document, which describes our company’s intentions and the methods which are related with validation. It can include information such as the scope of work, timeline, resources needed, risk assessment criteria, quality control procedures, and acceptance criteria. Web the technical storage or access is required to compose employee profiles to send advertising, or to track the. Web a free master validation plan (mvp) form to help medical device manufacturers with documenting a list of all company.Validation Master Plan (VMP) Downloadable Interactive Template.
Validation Master Plan
Example of a Validation Master Plan (VMP) Checklist Oriel STAT A
FREE 9+ Sample Validation Plan Templates in PDF MS Word
Validation Master Plan Template Validation Center
Validation Master Plan
Validation Master Plan Template Verification And Validation
Computerized System Validation Master Plan Free Word file download
FREE 9+ Sample Validation Plan Templates in PDF MS Word
Validation Master Plan
Web The Guidance Even Includes Templates For A Master Validation Plan, Iq, Oq, And Pq.
Web This Template Describes The Information That Needs To Be Presented In A Validation Master Plan And Provides Examples.
This Template Is A Tool For Creating A Customized Plan For Validating A Product, System, Or Process.
Web What Is A Validation Master Plan Template?
Related Post: