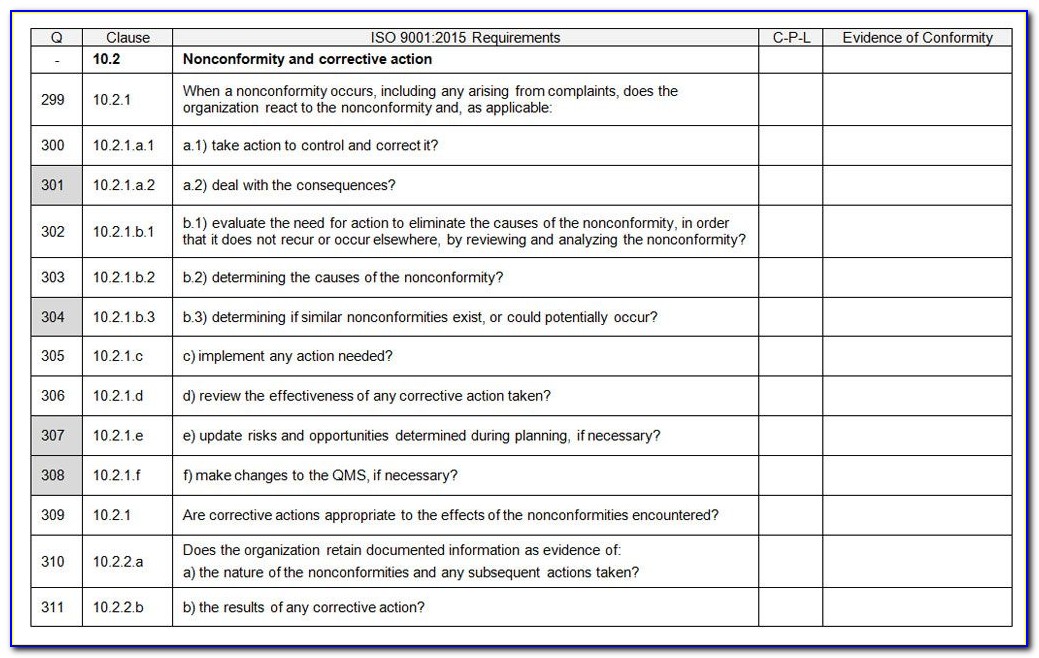
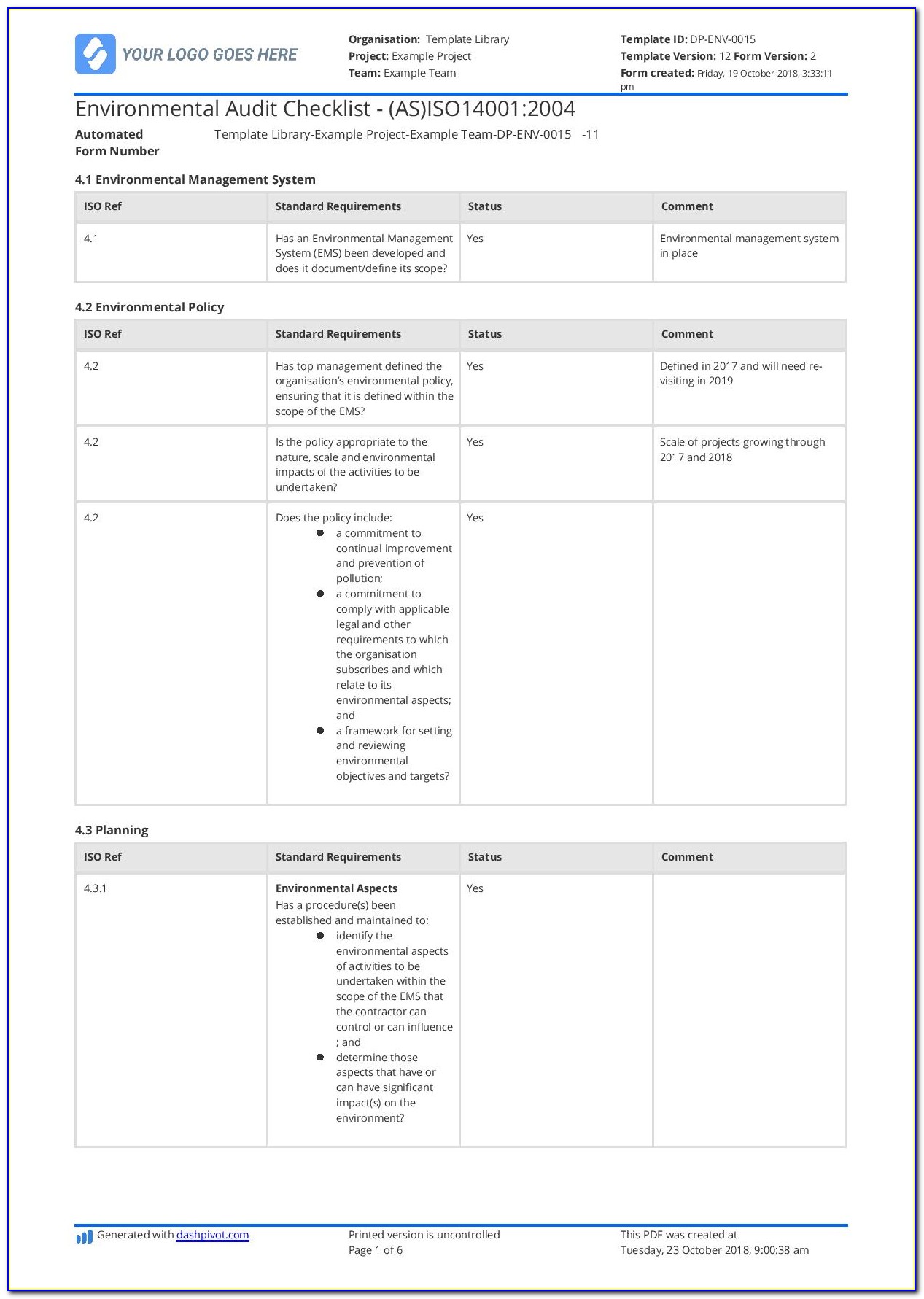
Iso 13485 Templates
Iso 13485 Templates - Web the iso 13485 is the standard for quality management in the medical device industry. Web in this article, you will find a quality manual template conforming to the requirements of regulation 2017/745 and. Web in accordance with both the fda qsr and the iso13485 law, before you can implement a qms system, including a form system,. Web from 1 january 2025, only iso 13485 issued by sac accredited certification bodies will be accepted for. Risk management plan the purpose of this document is to describe the risk. Web simply put, iso 13485 is a set of requirements defined by the international organization for standardization, designed. Web get latest iso 13485 templates for medical device from i3cglobal. Nowadays, there is much software providing diy templates. Here you can check the complete list of. Web iso 13485 document template: Here are all our posts on this standard, and. Iso 13485 compliance requires the implementation of various templates and documentation to. Web in this article, you will find a quality manual template conforming to the requirements of regulation 2017/745 and. Web our iso 13485:2016 procedures are designed for you to use with your iso 13485:2016 quality management system, as. Web. The document is fully editable so that you can adapt it to your company. Web iso 13485 document template: Web use our free iso 13485 procedure template and the list of iso 13485:2016 mandatory procedures to build your medical device quality system and get certified. Web iso 13485 document template: Iso 13485 compliance requires the implementation of various templates and. Web our iso 13485:2016 procedures are designed for you to use with your iso 13485:2016 quality management system, as. Here you can check the complete list of. Web simply put, iso 13485 is a set of requirements defined by the international organization for standardization, designed. Web download free management system templates for a range of standards including iso 9001 quality,. Web in this article, you will find a quality manual template conforming to the requirements of regulation 2017/745 and. Web the documentation template may be used for iso 13485 certification audit purposes. Iso 13485 compliance requires the implementation of various templates and documentation to. Web the pros and cons of the 4 best iso 13485 gap analysis templates. Web this. Web from 1 january 2025, only iso 13485 issued by sac accredited certification bodies will be accepted for. Web download free management system templates for a range of standards including iso 9001 quality, iso 14001. Web the pros and cons of the 4 best iso 13485 gap analysis templates. Web simply put, iso 13485 is a set of requirements defined. Iso 13485 compliance requires the implementation of various templates and documentation to. Web simply put, iso 13485 is a set of requirements defined by the international organization for standardization, designed. Web safetyculture checklists iso 13485 free iso 13485 audit checklists and pdf reports identify gaps in your quality. Web download free management system templates for a range of standards including. Web get latest iso 13485 templates for medical device from i3cglobal. Iso 13485 compliance requires the implementation of various templates and documentation to. Web the iso 13485 is the standard for quality management in the medical device industry. Web iso 13485 document template: Here are all our posts on this standard, and. Web download free management system templates for a range of standards including iso 9001 quality, iso 14001. Web iso 13485 document template: Web safetyculture checklists iso 13485 free iso 13485 audit checklists and pdf reports identify gaps in your quality. Web from 1 january 2025, only iso 13485 issued by sac accredited certification bodies will be accepted for. The document. Web in this article, you will find a quality manual template conforming to the requirements of regulation 2017/745 and. Web the pros and cons of the 4 best iso 13485 gap analysis templates. Web the iso 13485 is the standard for quality management in the medical device industry. The document is fully editable so that you can adapt it to. Web download free management system templates for a range of standards including iso 9001 quality, iso 14001. Web making templates — build and design forms. Quality plan the quality plan is the documented list of arrangements needed for the creation of. Web preview design and development file template. Web use our free iso 13485 procedure template and the list of. The document is fully editable so that you can adapt it to your company. Web the documentation template may be used for iso 13485 certification audit purposes. Web making templates — build and design forms. Web preview design and development file template. Iso 13485 compliance requires the implementation of various templates and documentation to. Risk management plan the purpose of this document is to describe the risk. Web our iso 13485:2016 procedures are designed for you to use with your iso 13485:2016 quality management system, as. Web download free management system templates for a range of standards including iso 9001 quality, iso 14001. Iso 13485:2016 is the international. Web check them out below. Checklist of mandatory documentation, description of. Web in accordance with both the fda qsr and the iso13485 law, before you can implement a qms system, including a form system,. Nowadays, there is much software providing diy templates. Web the pros and cons of the 4 best iso 13485 gap analysis templates. Web iso 13485 document template: Web the iso 13485 is the standard for quality management in the medical device industry. Here are all our posts on this standard, and. Web use our free iso 13485 procedure template and the list of iso 13485:2016 mandatory procedures to build your medical device quality system and get certified. Web download free eu mdr and iso 13485 pdf compliance materials: Web this table maps all requirements of the iso 13485:2016 (by section) to the relevant documents. Web safetyculture checklists iso 13485 free iso 13485 audit checklists and pdf reports identify gaps in your quality. Iso 13485:2016 is the international. Here are all our posts on this standard, and. Risk management plan the purpose of this document is to describe the risk. Checklist of mandatory documentation, description of. Web iso 13485 document template: Quality plan the quality plan is the documented list of arrangements needed for the creation of. Web simply put, iso 13485 is a set of requirements defined by the international organization for standardization, designed. Web this table maps all requirements of the iso 13485:2016 (by section) to the relevant documents. Iso 13485 compliance requires the implementation of various templates and documentation to. Web from 1 january 2025, only iso 13485 issued by sac accredited certification bodies will be accepted for. Web get latest iso 13485 templates for medical device from i3cglobal. The document is fully editable so that you can adapt it to your company. Web in accordance with both the fda qsr and the iso13485 law, before you can implement a qms system, including a form system,. Web the documentation template may be used for iso 13485 certification audit purposes. Web iso 13485 document template:Iso 13485 audit plan template beamluda
Iso 13485 Audit Report Sample
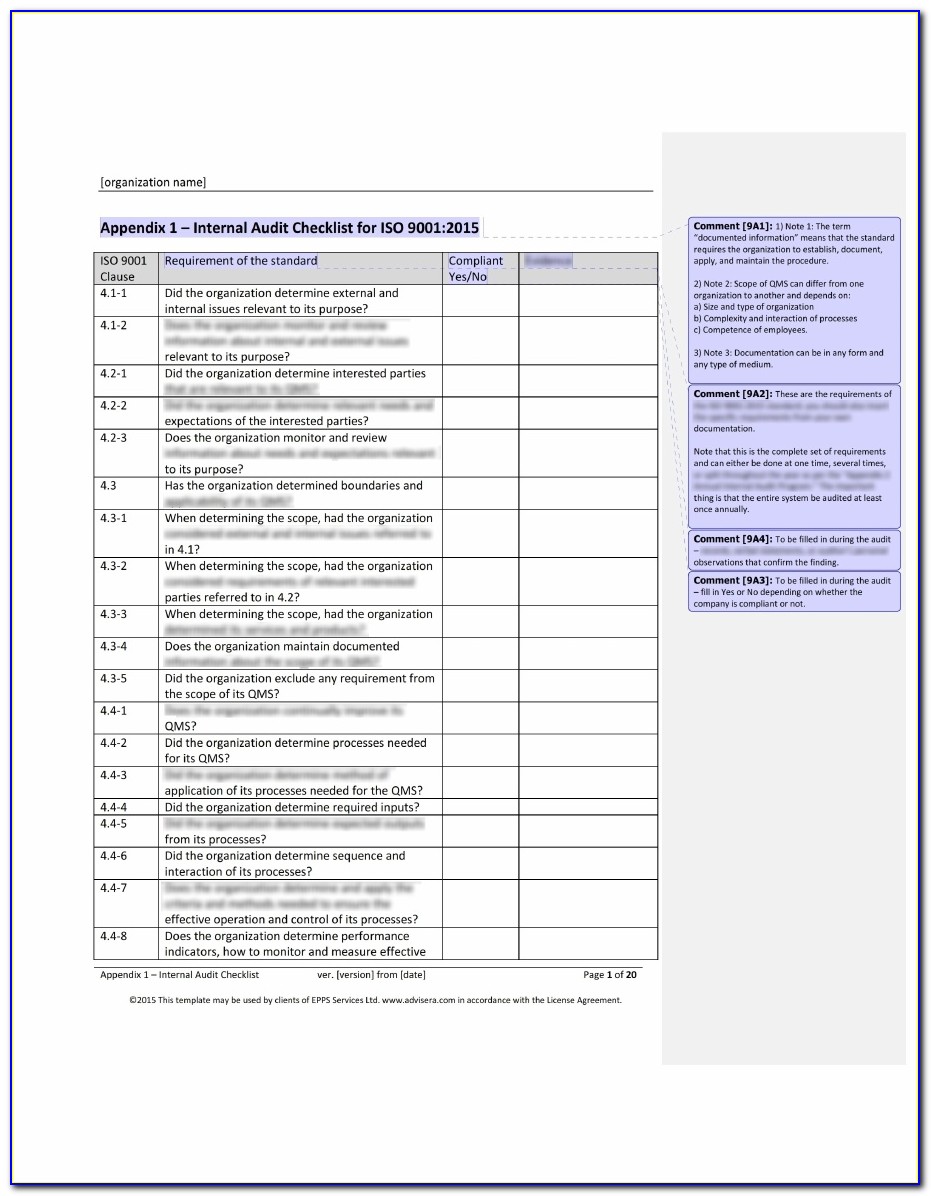
Iso 13485 Quality Manual Template Free
Iso 13485 Internal Audit Schedule Template
Iso 13485 Audit Plan Template
ISO 13485 Complaint Handling Forms
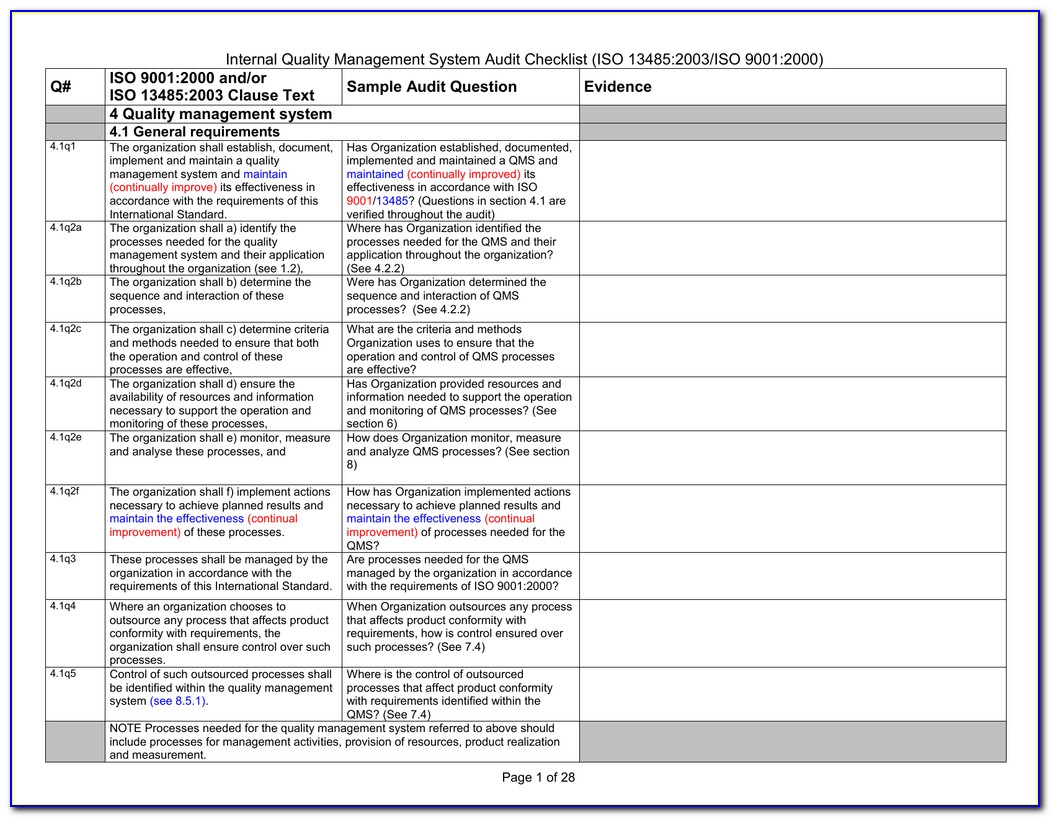
Iso 13485 Internal Audit Report Template
Best Tips ISO 13485 procedures with our free template (Version 2016)
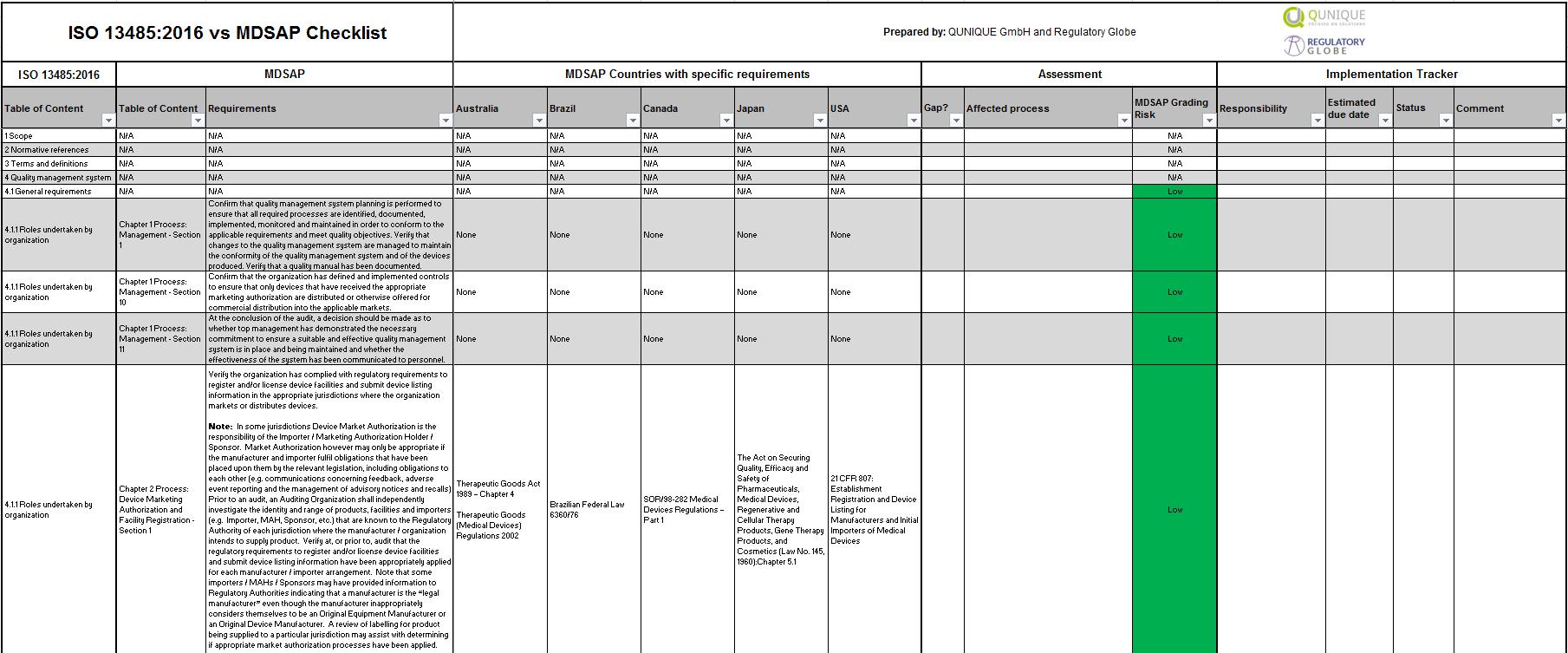
ISO 134852016 Internal Audit Toolkit
Iso 13485 Audit Report Template
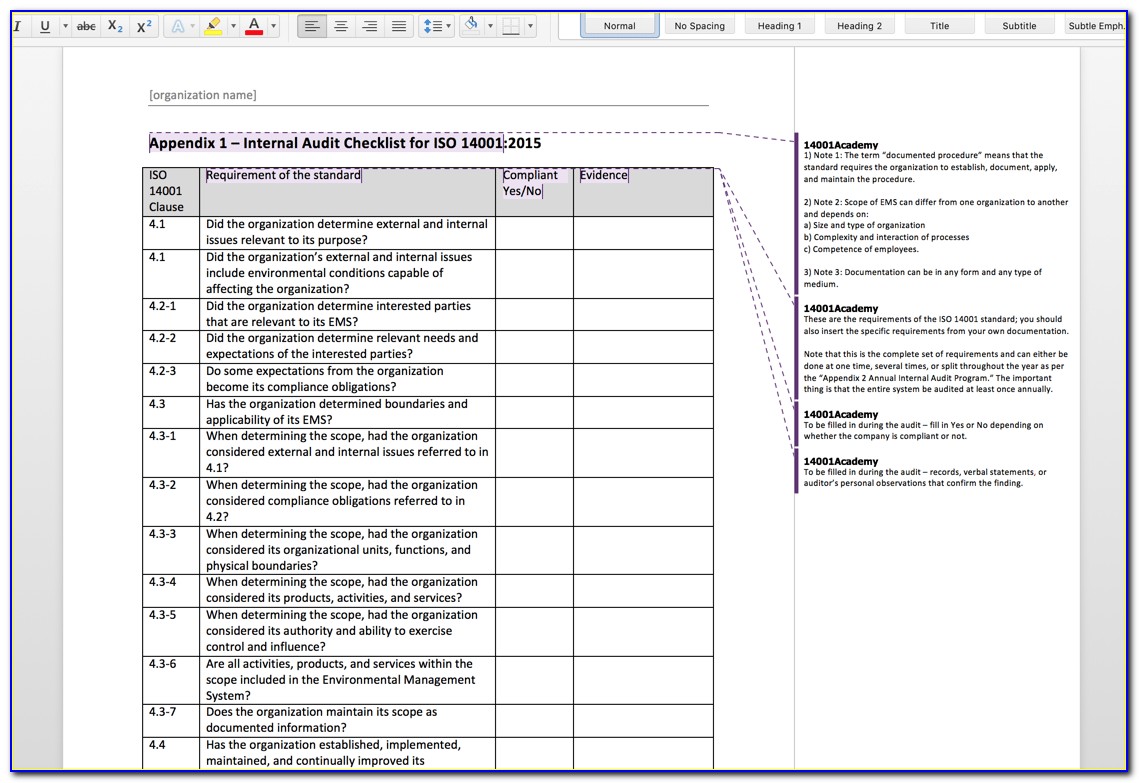
Web Making Templates — Build And Design Forms.
Web Preview Design And Development File Template.
Web Download Free Eu Mdr And Iso 13485 Pdf Compliance Materials:
Nowadays, There Is Much Software Providing Diy Templates.
Related Post: